High-Purity Recombinant Proteins for Biomedical Research
Exreprotein LLC delivers research-grade proteins and custom protein expression services built on advanced expression and purification technologies. Exreprotein LLC offers a growing portfolio of recombinant human cytokines and growth factors produced with verified biological activity and low endotoxin content for sensitive research applications.
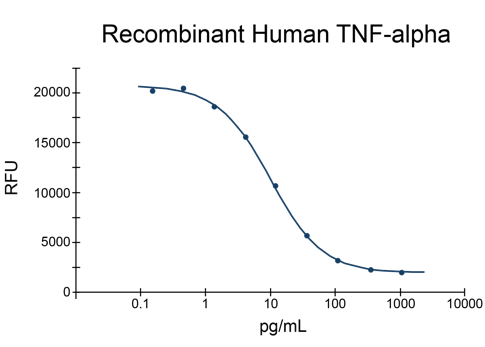
From foundational reagents such as bovine serum albumin and Human Serum Albumin to specialized growth factors including recombinant human HGF protein, Human Noggin, TNF-alpha, GDNF, and PDGF-BB, every product is engineered for purity, low endotoxin content, and traceable quality.
Product Filters
Explore Product Categories:
- Proteins (60)
- LIF Proteins (1)
- FGF-basic Proteins (1)
- TGF-beta 1 Proteins (1)
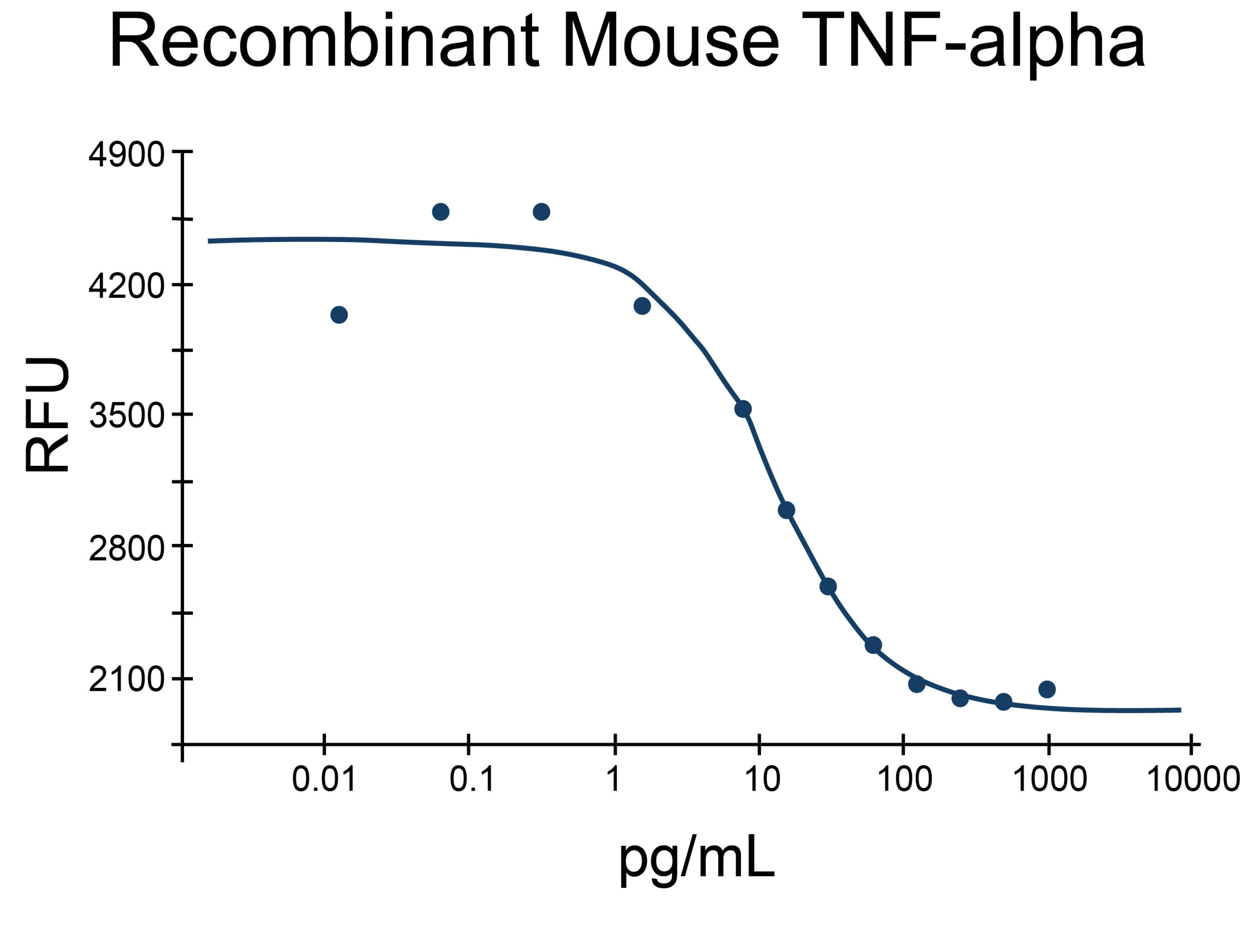
- TNF-alpha Proteins (2)
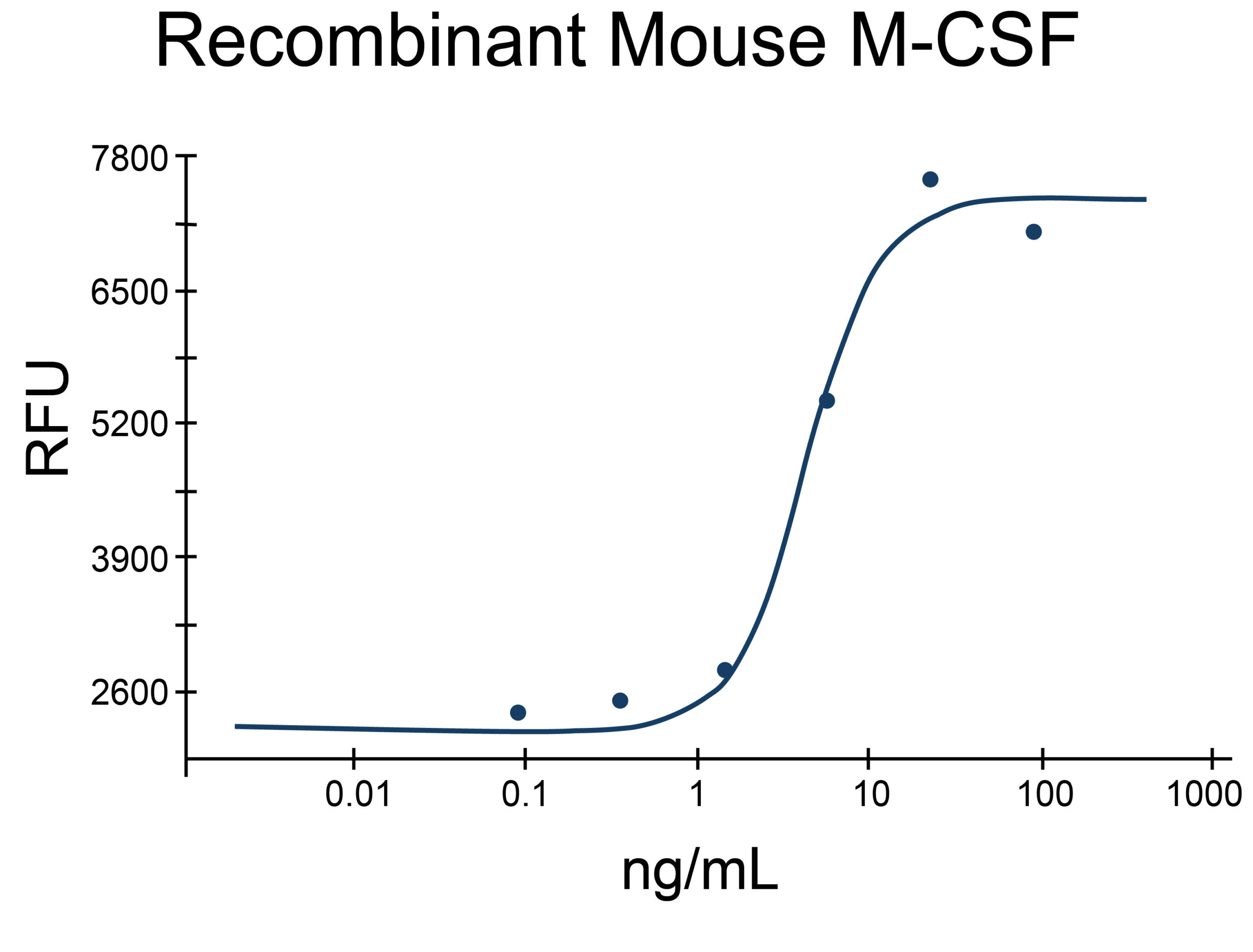
- M-CSF/CSF1 Proteins (2)
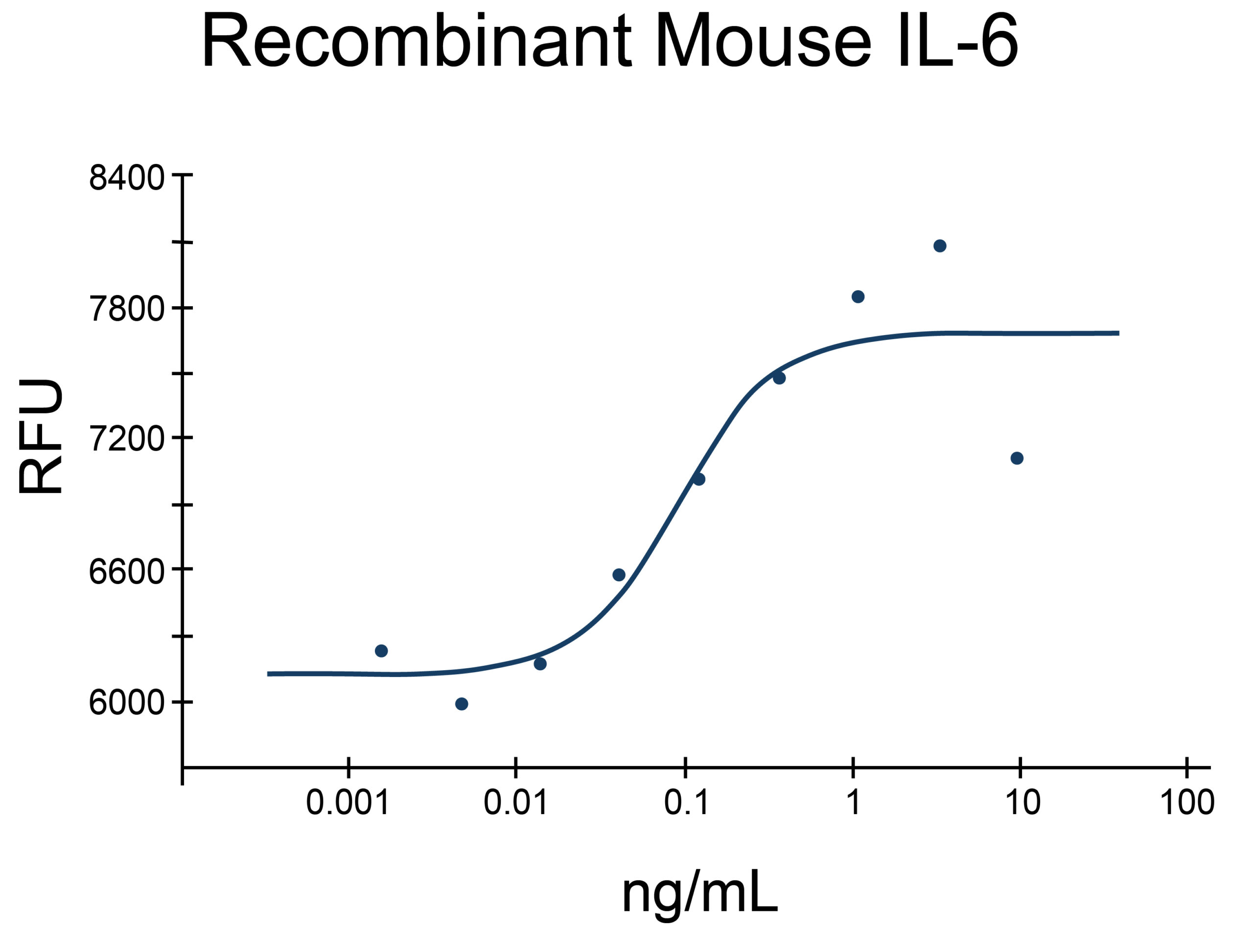
- IL-6 Proteins (5)
- EGF Proteins (1)
- KGF/FGF-7 Proteins (1)
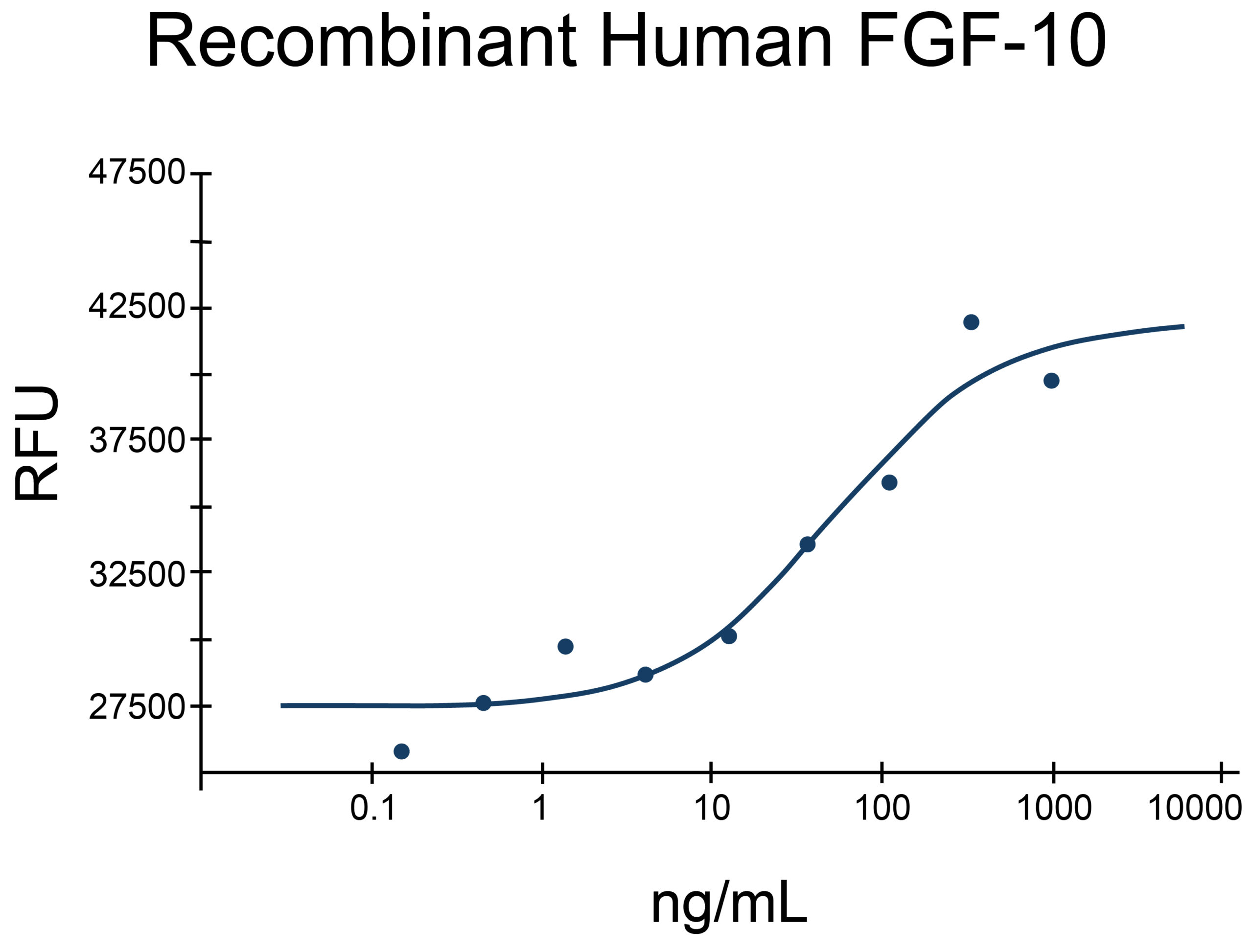
- FGF-10 Proteins (1)
- IFN-gamma Proteins (2)
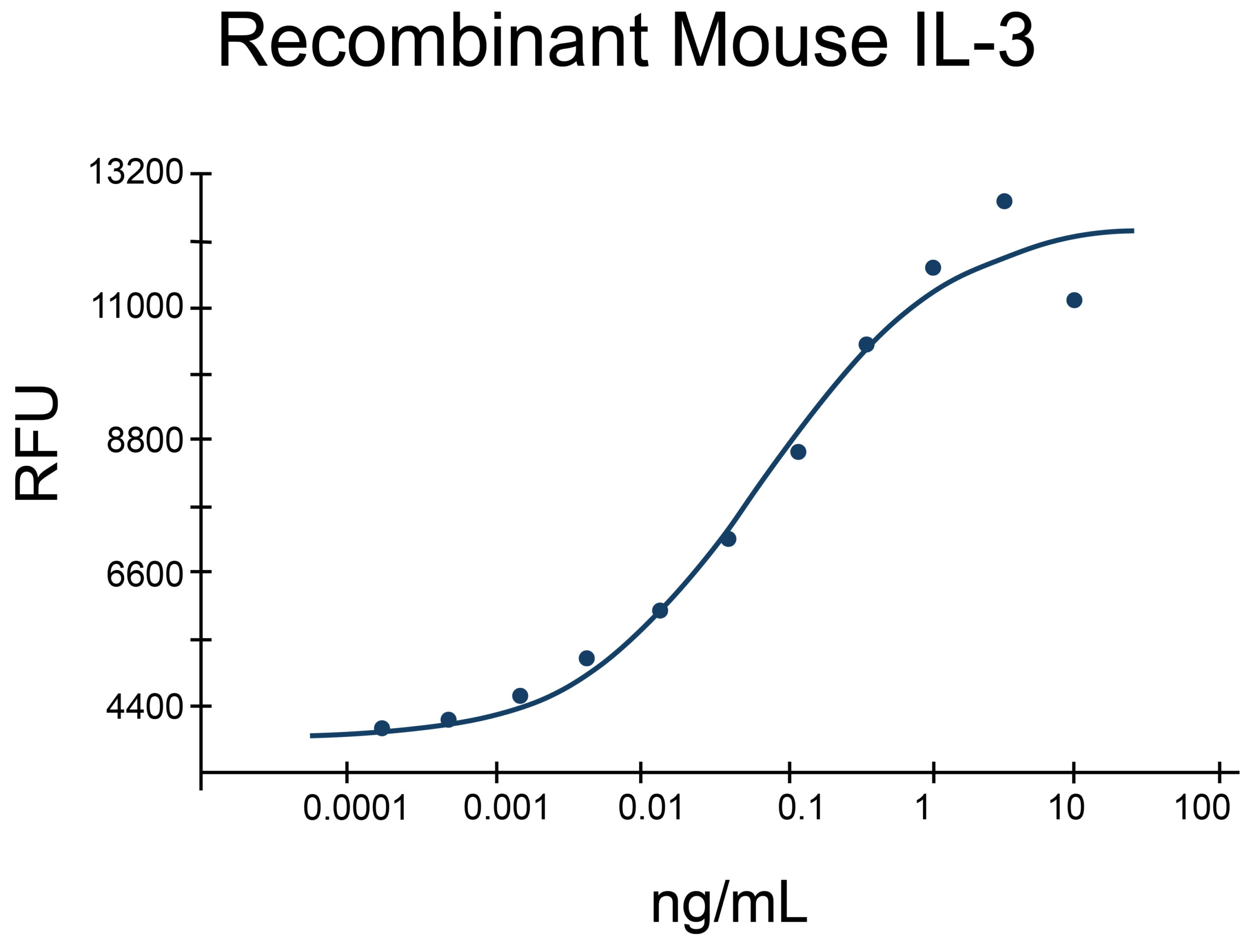
- IL-3 Proteins (2)
- R-Spondin 1/RSPO1 Proteins (1)
- Noggin/NOG Proteins (2)
- GM-CSF/CSF2 Proteins (2)
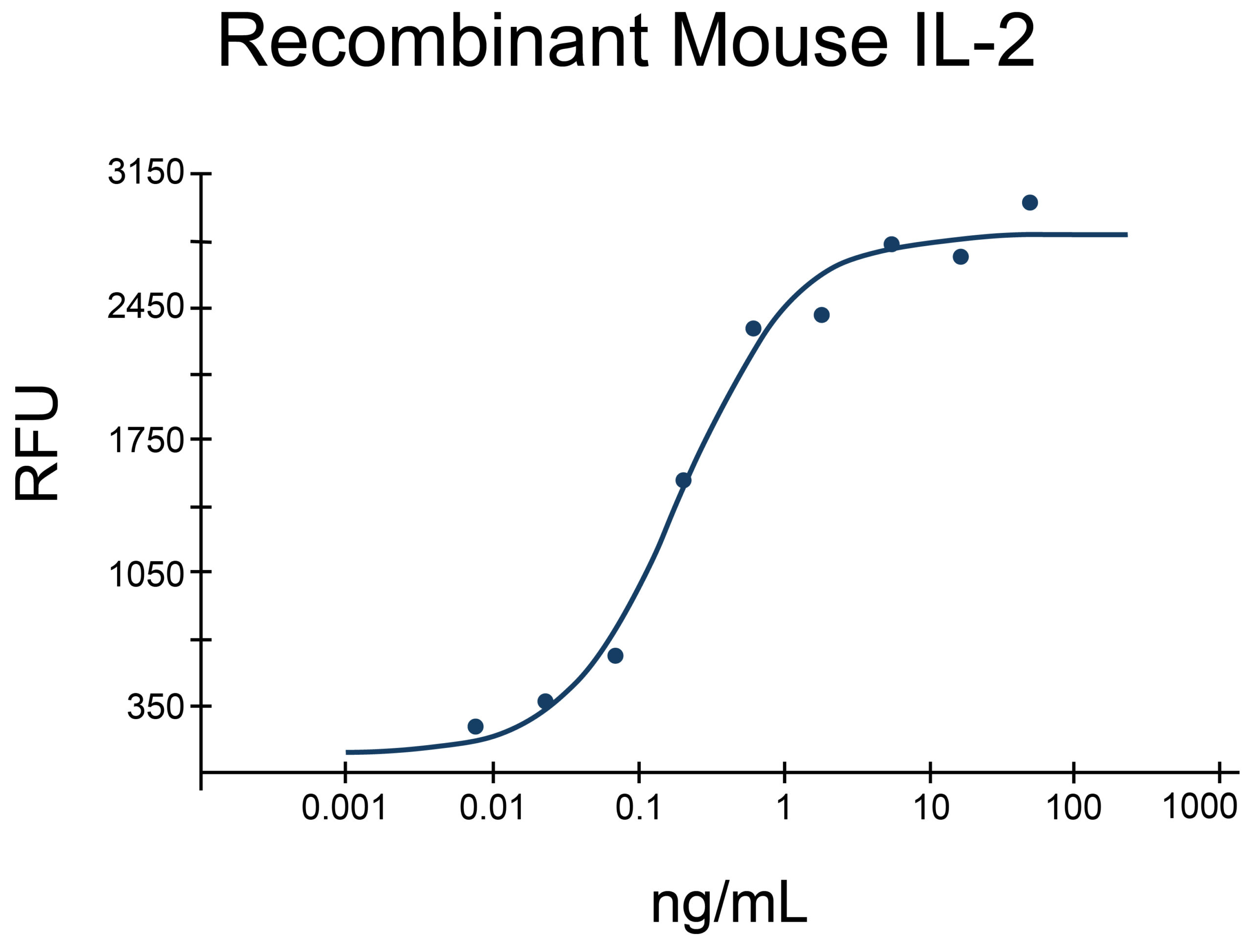
- IL-2 Proteins (2)
- IL-15 Proteins (1)
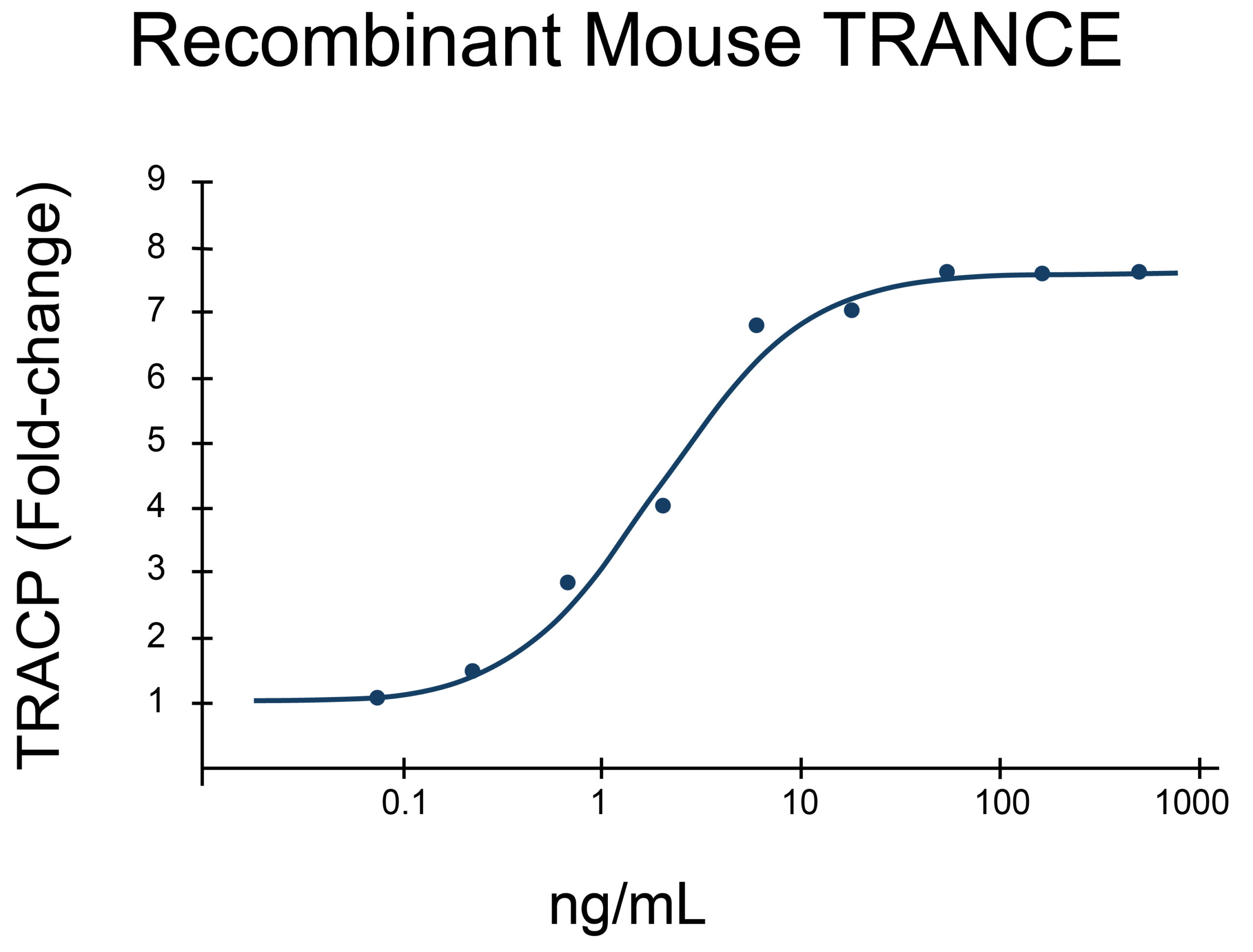
- TRANCE/RANKL Proteins (1)
- IL-1 beta/IL-1F2 Proteins (1)
- GDNF Proteins (1)
- PDGF-BB Proteins (1)
- Thrombopoietin/TPO Proteins (2)
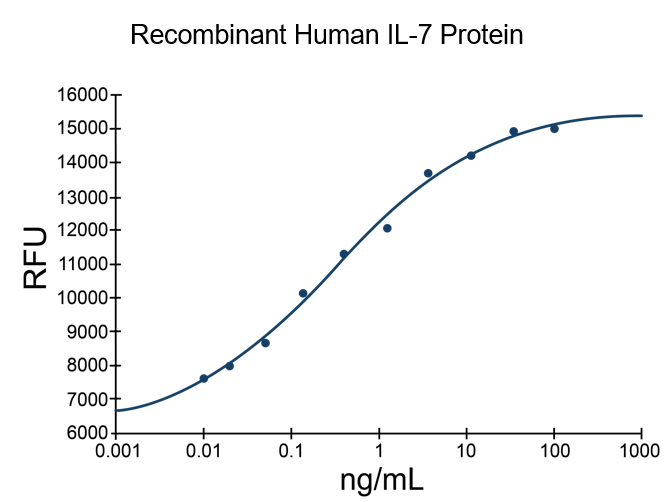
- IL-7 Proteins (1)
- IL-21 Proteins (1)
- BMP-4 Proteins (1)
- Activin A Proteins (1)
- IL-4 Proteins (2)
- HGF Proteins (1)
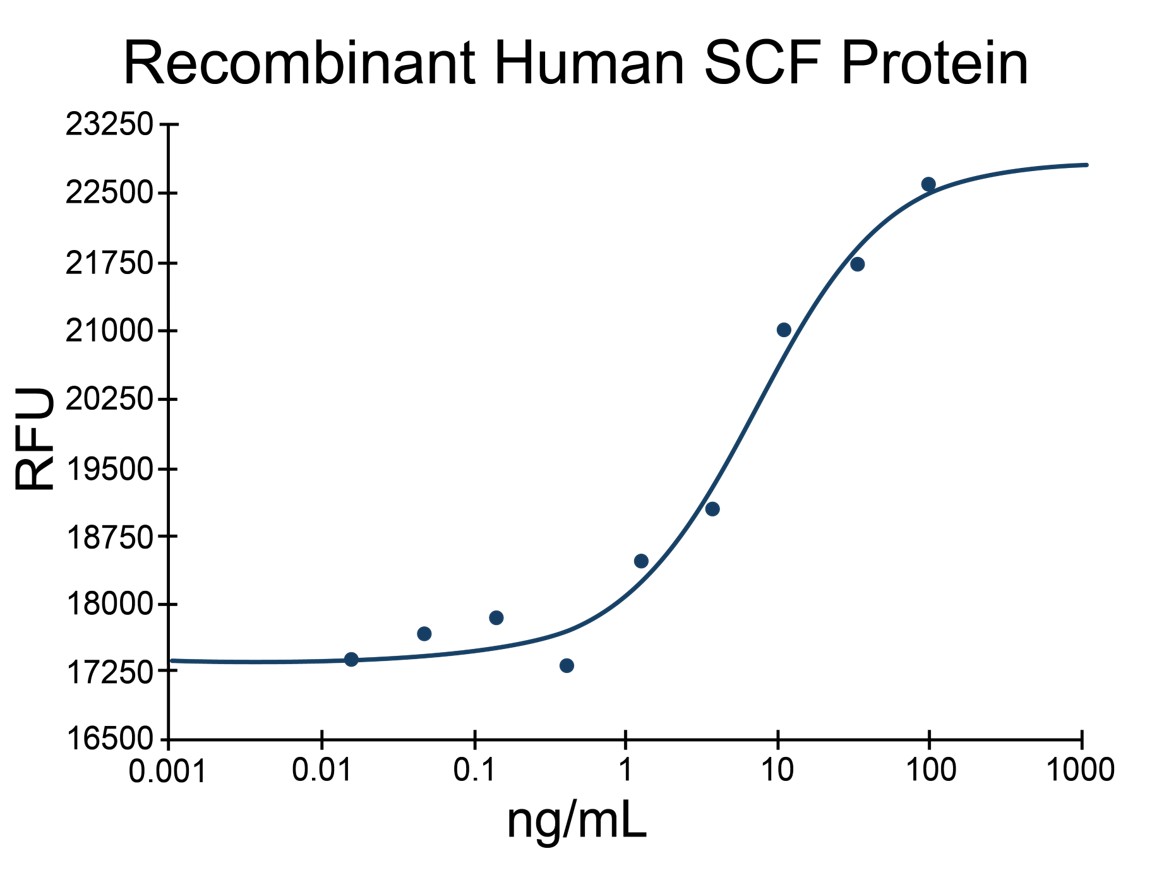
- SCF Proteins (2)
- VEGF Proteins (1)
- R-Spondin 3/RSPO3 Proteins (1)
- BDNF Proteins (1)
- Flt-3 Ligand/FLT3L Proteins (2)
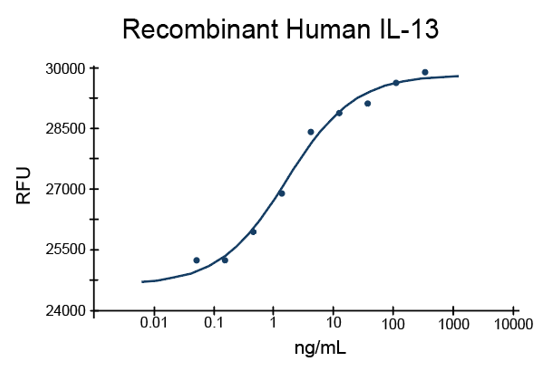
- IL-13 Proteins (1)
- Neurotrophin 3/NT-3 Proteins (1)
- Epiregulin Proteins (1)
- BMP-2 Proteins (1)
- Heregulin beta1 Proteins (1)
- PDGF-AA Proteins (1)
- G-CSF Proteins (1)
- GDF-15 Proteins (1)
- Serum Albumin Proteins (4)
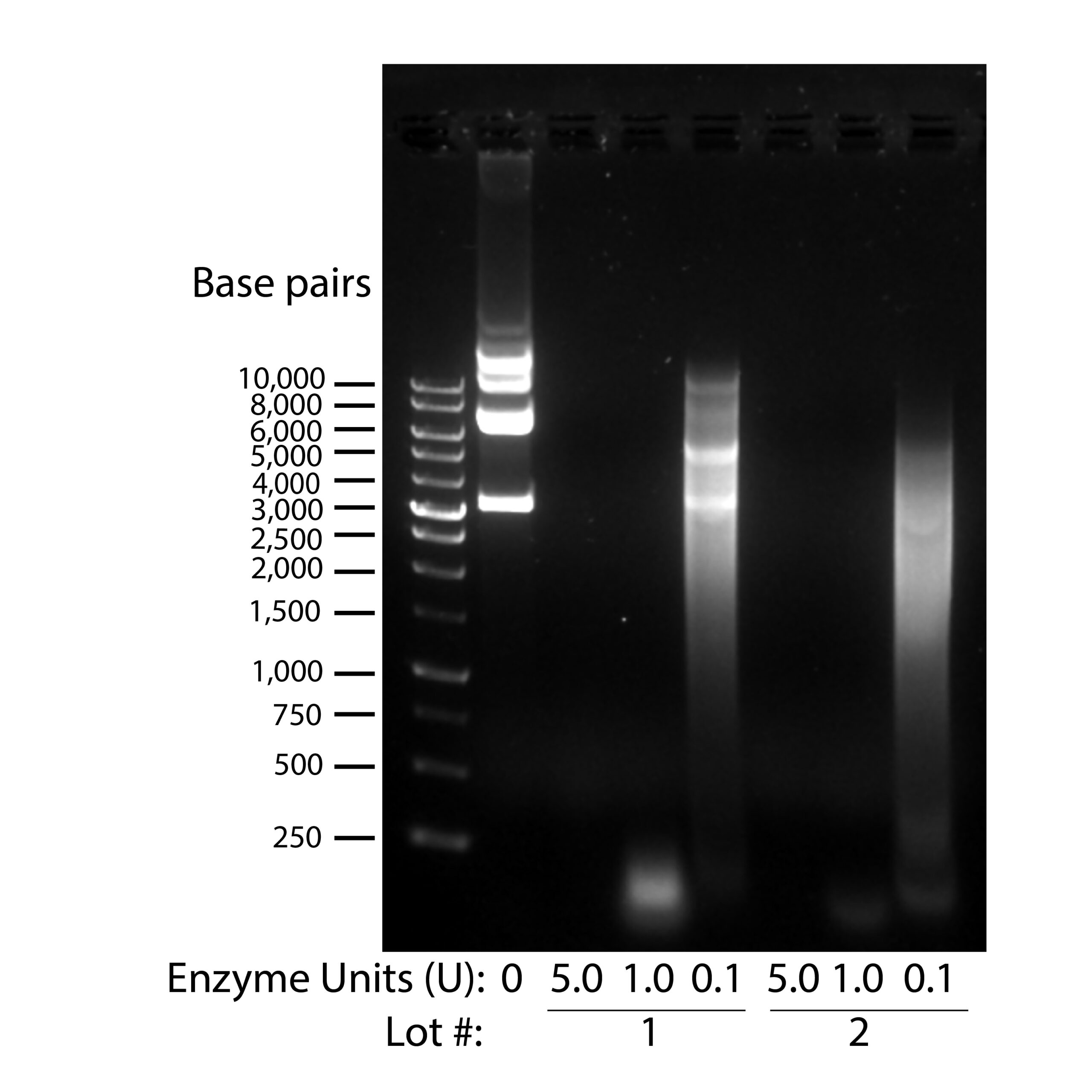
- ExreNuclease/NucA Enzyme (1)
- ELISAs/Assays (54)
- IFN-gamma Assays (6)
- IL-2 Assays (2)
- IL-6 Assays (2)
- IL-8 Assays (1)
- IL-10 Assays (3)
- IL-12p70 Assays (1)
- IL-12/IL-23p40 Assays (1)
- IL-17A Assays (2)
- sIL-2R Assays (1)
- CD106/sVCAM-1 Assays (1)
- Adiponectin /Acrp30 Assays (1)
- CRP Assays (1)
- TGF-beta 1 Assays (1)
- TNF-alpha Assays (3)
- EGF Assays (1)
- VEGF-A Assays (1)
- MCP-1 Assays (1)
- IP-10 Assays (1)
- FGF-basic Assays (1)
- HGF Assays (1)
- MMP-2 Assays (1)
- IL-18 Assays (1)
- IL-1 beta Assays (2)
- IFN-alpha Assays (1)
- Resistin Assays (1)
- MIP-1 alpha Assay (2)
- TARC Assays (1)
- Granzyme B Assays (1)
- IL-4 Assays (3)
- IL-1 alpha Assays (1)
- IL-5 Assays (1)
- IL-9 Assays (1)
- IL-13 Assays (1)
- IL-17F Assays (1)
- IL-21 Assays (1)
- IL-22 Assays (1)
- GM-CSF Assays (1)
- IL-23 Assays (1)
- G-CSF Assays (1)
- Cell Culture (9)
- Western blot/Electrophoresis (19)
- Antibodies (17)
- Buffers & Reagents (12)
- Molecular Biology (56)
Products
Showing 29–56 of 60 results
How We Evaluate Recombinant Protein Quality
Not all recombinant proteins meet the same quality standards. Proper evaluation is essential
for reproducible and biologically meaningful results.
- Purity and IdentityHigh-quality recombinant proteins should meet:
- ≥97% purity by SDS-PAGE and/or HPLC
- Confirmed identity by mass spectrometry or peptide mapping
- Endotoxin ContentEndotoxins can alter cell behavior and activate inflammatory pathways. Recommended limits include:
- <0.1 EU/µg for general in vitro research
- <0.01 EU/µg for highly sensitive cell-based assays
- Biological ActivityFunctional validation may include:
- Receptor binding assays
- Cell-based signaling or proliferation assays
- Protein-specific functional readouts
- Optimized Lyophilization and Product TraceabilityExreprotein LLC proteins are commonly supplied in optimized lyophilized formats to ensure:
- Long-term stability
- Preserved biological activity
- Consistent reconstitution
- Simplified storage and shipping
Custom Protein Expression and Purification Services
In addition to catalog proteins, Exreprotein LLC provides custom protein expression services
using the same platforms applied to our research-grade products.
- Custom Expression Capabilities
- Codon optimization and gene synthesis
- Expression in bacterial, yeast, insect, or mammalian systems
- Native or engineered constructs
- Tag-free or cleavable-tag designs
- Custom Purification and Quality Targets
- Multi-step purification strategies
- Defined endotoxin specifications
- Host cell protein and residual DNA clearance
- Protein-specific formulation and lyophilization